THE NEED.
Microfluidics technology has revolutionised key applications like drug development, stem cell research, microbiological analysis, medical diagnosis, personalised medicine and chemical biology, just to name but a few. Microfluidic systems are always more present in chemistry and biochemistry labs, driving the development of new components and processes for the injection, mixing, pumping, and storing of fluids in the microchannels.
In this way, the progress of microfluidics technology has opened a completely new market for Lab-On-Chip systems (LOC), which are miniaturised devices intended to replicate what happens in a real lab, drastically improving cost efficiency, parallelization, ergonomics, diagnostic speed and sensitivity.
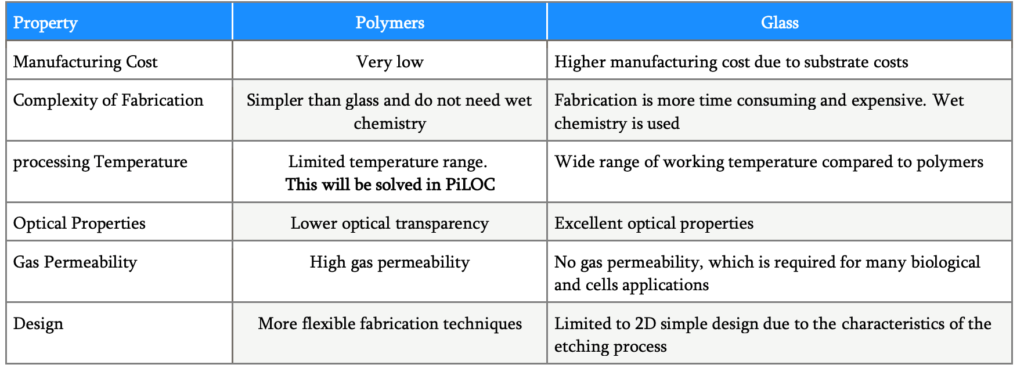
Considering that the largest majority of microfluidic chips is made of glass or silicon due to the mature manufacturing process and excellent optical properties, surface stability, solvent compatibility, one question is puzzling the LOC industry: how to deal with the increased complexity of systems?
In fact, the need for high speed, flexible, multiparametric systems are challenging the technological and cost constraints. Indeed, an increasing number of discrete components like actuators and valves can be implemented on glass, but who is willing to spend up to 500€ for a disposable chip?
The latter is the driving force towards the research of LOC based on polymer chips, that not only have most advantages of glass chips like transparency, electrical insulation, biocompatibility but are fast and cost effective to fabricate in large volumes.
Unfortunately, while microfluidic channels can be easily manufactured with polymers, active components like actuators and valves integration are an unsolved issue, as their full integration with polymers would take place at processing temperature far beyond the melting point of polymers, and the only way to have such components in a LOC is by gluing them, which is totally unreproducible and leading to unreliable outcomes.
THE CHALLENGE.
Microfluidics technology has revolutionised key applications like drug development, stem cell research, microbiological analysis, medical diagnosis, personalised medicine and chemical biology, just to name but a few.
Microfluidic systems are ever more present in chemistry and biochemistry labs, driving the development of new components and processes for the injection, mixing, pumping, and storing of fluids in the microchannels.
As such, the progress of microfluidics technology has opened a completely new market for Lab-On-Chip systems (LOC), which are miniaturised devices intended to replicate experiments normally carried out in a real lab, drastically improving cost efficiency, parallelization, ergonomics, diagnostic speed and sensitivity.
Using the piezoelectric effect in microfluidics is not recent. There are several publications in which a piezoelectric bulk ceramic is glued to a microfluidic chip.1 The problem with such an approach is that the performance of the system is not reproducible and depends on the gluing quality. In addition, bulk ceramics need high driving voltages, which is a critical disadvantage compared to non-piezo-based components. Both above-mentioned limitations will be resolved by using piezoelectric thin films deposited directly on microfluidic chips.